
Content Curator
SRMJEEE 2018 Question paper with answer key pdf is available for download. The exam was successfully organized by SRM University. The question paper comprised a total of 140 questions.
SRMJEEE 2018 Question Paper with Answer Key PDFs in English
| SRMJEEE 2018 Question Paper PDF | SRMJEEE 2018 Answer Key PDF |
|---|---|
| Download PDF | Download PDF |


































SRMJEEE Previous Year Question Paper with Answer Key PDFs
Similar Exam Question Papers
SRMJEEE Questions
1. The molar conductivities at infinite dilution for Na2SO4,K2S04,KCl, HCl and HCOONa at 300K are 260, 308, 150, 426, and 105 S cm2 mol-1, respectively. What will be A+m for formic acid in the same unit?
The molar conductivities at infinite dilution for Na2SO4,K2S04,KCl, HCl and HCOONa at 300K are 260, 308, 150, 426, and 105 S cm2 mol-1, respectively. What will be A+m for formic acid in the same unit?
429
405
531
381
2. A precipitate is formed if the ionic product is __________?
A precipitate is formed if the ionic product is __________?
Greater than the solubility product
Lesser than the solubility product
Equal to solubility product
Independent of solubility product
3. If VA and VVB are the potentials of the two points A and B, what will be the correct relation between the electric field and the potential difference between the points? The distance between the points is d.
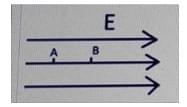

- VA- VB = \(\frac{d}{E}\)
- VA-VB = Ed
- VB- VA = Ed
- VA-VA = \(\frac{E}{d}\)
4. Electrophilic halogenation of phenol does not require catalyst because
Electrophilic halogenation of phenol does not require catalyst because
Phenol is electron rich
Oxygen in phenol is more reactive
Phenol is electron deficient as oxygen is the second is the second most electronegative element
Phenol is a planar molecule
5. Following reaction can produce three products. The order of their amount is
Following reaction can produce three products. The order of their amount is
A> C> B
C> B> A
A> B> C
C> A> B
6. A concave mirror of radius 30 cm is placed in water. It’s focal length in air and water differ by
- 15
- 20
- 30
- 0



.png?h=160&w=320&mode=stretch)
.png?h=160&w=320&mode=stretch)
.png?h=160&w=320&mode=stretch)
.png?h=160&w=320&mode=stretch)



Comments