Question:
Statement-1: Sublimation is a purification technique that is used to separate those solid substances which change from solid to vapor state without passing through liquid state.
Statement-2: If external atmospheric pressure is reduced, then boiling point of substance decreases.
Which of the following is correct?
Statement-1: Sublimation is a purification technique that is used to separate those solid substances which change from solid to vapor state without passing through liquid state.
Statement-2: If external atmospheric pressure is reduced, then boiling point of substance decreases.
Which of the following is correct?
Statement-2: If external atmospheric pressure is reduced, then boiling point of substance decreases.
Which of the following is correct?
Show Hint
Sublimation is used for purifying volatile substances, while changes in pressure can affect the boiling point of substances, which is particularly important in distillation and other purification techniques.
Updated On: Jan 28, 2026
- Statement-1 and Statement-2 both are correct.
- Statement-1 and Statement-2 both are incorrect.
- Statement-1 is correct but Statement-2 is incorrect.
- Statement-1 is incorrect but Statement-2 is correct.
Hide Solution
Verified By Collegedunia
The Correct Option is A
Solution and Explanation
Step 1: Sublimation.
Sublimation is indeed a purification process used to separate substances that change directly from solid to gas without going through the liquid phase. This is a common process for certain solids like iodine and dry ice.
Step 2: Boiling point and pressure.
The boiling point of a substance is influenced by the atmospheric pressure. If the pressure is lowered, the boiling point decreases. This is because less energy is required for the substance to vaporize.
Step 3: Conclusion.
Both statements are correct, so the correct answer is (1).
Sublimation is indeed a purification process used to separate substances that change directly from solid to gas without going through the liquid phase. This is a common process for certain solids like iodine and dry ice.
Step 2: Boiling point and pressure.
The boiling point of a substance is influenced by the atmospheric pressure. If the pressure is lowered, the boiling point decreases. This is because less energy is required for the substance to vaporize.
Step 3: Conclusion.
Both statements are correct, so the correct answer is (1).
Was this answer helpful?
0
0
Top JEE Main Chemistry Questions
- C5H10 \(\xrightarrow{h\nu}\) possible structural isomers?
- The number of isoelectronic species among S2−, C4−, Mn2+, Co3+ and Fe3+ is ‘n’.
If ‘n’ moles of AgCl is formed during the reaction of the complex with formula
CoCl2(en)2NH3
with excess AgNO3 solution, then the number of electrons present in the t2g orbital of the complex is ________.- JEE Main - 2026
- Chemistry
- Organic Chemistry
Write the correct order of rate of reaction of following with PhN$_2$Cl

- JEE Main - 2026
- Chemistry
- Organic Reactions
- K$_{sp}$ of Ag$_2$CrO$_4$ = 32x
K$_{sp}$ of AgBr = 4y Then, the ratio of molarity (solubility) of (1) to (2) is:
- JEE Main - 2026
- Chemistry
- Ionic Equilibrium In Solution
- For first order reaction, rate constant at 27°C and t°C is $1.5 \times 10^3$ and $4.5 \times 10^3$ respectively. If the activation energy of the reaction is 60 kJ, then find the temperature $t$.
- JEE Main - 2026
- Chemistry
- Chemical Kinetics
View More Questions
Top JEE Main States of matter Questions
- Given below are two statements: Statement I: Sublimation is a purification technique that is used to separate those solid substances which change from solid to vapour state without passing through liquid state. Statement II: If external atmospheric pressure is reduced, then boiling point of a substance is decreased. In the light of the above statements, choose the correct option.
- JEE Main - 2026
- Chemistry
- States of matter
- Based on the given figure, the number of correct statement/s is/are _______
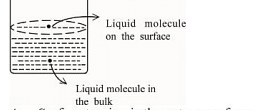
A. Surface tension is the outcome of equal attractive and repulsive forces acting on the liquid molecule in bulk
B. Surface tension is due to uneven forces acting on the molecules present on the surface
C. The molecule in the bulk can never come to the liquid surface
D. The molecules on the surface are responsible for vapour pressure if the system is a closed system- JEE Main - 2023
- Chemistry
- States of matter
- Number of molecules and moles in 2.8375 litre of O2 at STP.
- JEE Main - 2023
- Chemistry
- States of matter
- A metal chloride contains 55% by mass of chlorine. 100 mL of vapours gives 0.57 gm of chlorine at STP. Calculate the molecular mass of metal chloride. (Nearest integer)
- JEE Main - 2023
- Chemistry
- States of matter
Which one of the following is the correct PV vs P plot at constant temperature for an ideal gas ? (P and V stand for pressure and volume of the gas respectively)

- JEE Main - 2021
- Chemistry
- States of matter
View More Questions
Top JEE Main Questions
If stress at $x = \ell/3$ from bottom is $\frac{W}{A} + \frac{2}{\gamma} \frac{w}{A}$ then find $\gamma$ :

- JEE Main - 2026
- Dimensional analysis
- If $\vec{r} = 10t^2 \hat{i} + 5t^3 \hat{j}$ and mass of object, $m = 0.1 \text{ kg}$ then at $t = 1 \text{ sec}$ :-
(A) momentum = $2\hat{i} + 1.5\hat{j}$
(B) force = $2\hat{i} + 3\hat{j}$
(C) Angular momentum = $5\hat{k}$
(D) Torque = $20\hat{k}$- JEE Main - 2026
- Dimensional analysis
- If angular position of a particle is given by $\theta = \frac{t^4}{4} + t^2$. Find angular acceleration at $t = 1s$:
- JEE Main - 2026
- Dimensional analysis
- The dimension of $\frac{1}{2} \epsilon_0 E^2$ is $M^a L^b T^c$ then value of $a - 2b + c = ?$
- JEE Main - 2026
- Dimensional analysis
- Let \( f(x) = \int \frac{(2 - x^2) \cdot e^x \cdot \sqrt{1 + x} \cdot (1 - x)^{3/2}}{dx} \). If \( f(0) = 0 \), then \( f\left(\frac{1}{2}\right) \) is equal to:
View More Questions