Which one of the following has the same number of atoms as are in 6g of H2O
Which one of the following has the same number of atoms as are in 6g of H2O
0.4G He
22g CO2
1g H2
12g CO
The Correct Option is C
Solution and Explanation
The correct option is: (C): 1g H2.
Avogadro's number corresponds to the quantity of atoms or molecules found within 1 mole of any substance, and it is expressed as 6.022 x 10^23 atoms/mol. Molar mass signifies the weight of 1 mole of a substance.
Top Questions on Law Of Chemical Equilibrium And Equilibrium Constant
- When potassium iodide is added to an aqueous solution of potassium ferricyanide, a reversible reaction is observed in which a complex P is formed. In a strong acidic medium, the equilibrium shifts completely towards P. Addition of zinc chloride to P in a slightly acidic medium results in a sparingly soluble complex Q.
- JEE Advanced - 2024
- Chemistry
- Law Of Chemical Equilibrium And Equilibrium Constant
Which of the following does not form a buffer solution?
- TS EAMCET - 2023
- Chemistry
- Law Of Chemical Equilibrium And Equilibrium Constant
- (i) $ X ( g ) \rightleftharpoons Y ( g )+ Z$ (g) $ K _{ p 1}=3$ (ii) $A ( g ) \rightleftharpoons 2 B ( g )\,\, K _{ p 2}=1$ If the degree of dissociation and initial concentration of both the reactants $X ( g )$ and $A ( g )$ are equal, then the ratio of the total pressure at equilibrium $\left(\frac{p_1}{p_2}\right)$ is equal to $x : 1$ The value of $x$ is _________ (Nearest integer)
- JEE Main - 2023
- Chemistry
- Law Of Chemical Equilibrium And Equilibrium Constant
- At 500 K, for a reversible reaction \(A_{2_{(g)}}+B_{2_{(g)}}⇌2AB_{(g)}\) in a closed container, KC = 2 × 10-5. In the presence of catalyst, the equilibrium is attaining 10 times faster. The equilibrium constant KC in the presence of catalyst at the same temperature is
- KCET - 2023
- Chemistry
- Law Of Chemical Equilibrium And Equilibrium Constant
- Given below are two reactions, involved in the commercial production of dihydrogen (H2). The two reactions are carried out at temperature “T1 ” and “T2 ” respectively
\(C(S)+H_2O(g)\underrightarrow {T_1}\;CO(g)+H_2(g)\)
\(CO(g)+H_2O(g)\underrightarrow{T_2}\;CO_2(g)+H_2(g)\)
The temperature \(T_1\) and \(T_2\) are correctly related as- JEE Main - 2023
- Chemistry
- Law Of Chemical Equilibrium And Equilibrium Constant
Questions Asked in TS EAMCET exam
The roots of the equation x4 + x3 - 4x2 + x + 1 = 0 are diminished by h so that the transformed equation does not contain x2 term. If the values of such h are α and β, then 12(α - β)2 =
- TS EAMCET - 2023
- Quadratic Equations
The number of electrons with (n+1) values equal to 3,4 and 5 in an element with atomic number (z) 24 are respectively (n = principal quantum number and l = azimuthal quantum number)
- TS EAMCET - 2023
- atom structure models
Two convex lenses of focal lengths 20 cm and 30 cm are placed in contact with each other co-axially. The focal length of the combination is:
- TS EAMCET - 2023
- Ray optics and optical instruments
- If the ratio of densities of two substances is 5:6 and the ratio of their specific heat capacities is 3:5, then the ratio of heat energies required per unit volume so that the two substances can have same temperature rise is:
- TS EAMCET - 2023
- Thermodynamics
If i=√-1 then
\[Arg\left[ \frac{(1+i)^{2025}}{1+i^{2022}} \right] =\]- TS EAMCET - 2023
- Trigonometric Identities
Concepts Used:
Law of Chemical Equilibrium
Law of Chemical Equilibrium states that at a constant temperature, the rate of a chemical reaction is directly proportional to the product of the molar concentrations of the reactants each raised to a power equal to the corresponding stoichiometric coefficients as represented by the balanced chemical equation.
Let us consider a general reversible reaction;
A+B ↔ C+D
After some time, there is a reduction in reactants A and B and an accumulation of the products C and D. As a result, the rate of the forward reaction decreases and that of backward reaction increases.
Eventually, the two reactions occur at the same rate and a state of equilibrium is attained.
By applying the Law of Mass Action;
The rate of forward reaction;
Rf = Kf [A]a [B]b
The rate of backward reaction;
Rb = Kb [C]c [D]d
Where,
[A], [B], [C] and [D] are the concentrations of A, B, C and D at equilibrium respectively.
a, b, c, and d are the stoichiometric coefficients of A, B, C and D respectively.
Kf and Kb are the rate constants of forward and backward reactions.
However, at equilibrium,
Rate of forward reaction = Rate of backward reaction.
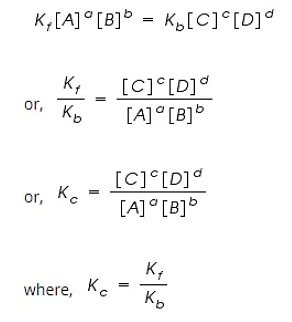
Kc is called the equilibrium constant expressed in terms of molar concentrations.
The above equation is known as the equation of Law of Chemical Equilibrium.



