The maximum number of possible isomers (including stereoisomers) which may be formed on mono-bromination of 1-methylcyclohex-1-ene using Br2 and UV light is ___.
The maximum number of possible isomers (including stereoisomers) which may be formed on mono-bromination of 1-methylcyclohex-1-ene using Br2 and UV light is ___.
Correct Answer: 13
Approach Solution - 1

The monobromination of 1-methylcyclohexene under UV light occurs via a free radical mechanism. This process generates allyl radicals, which are stabilized through resonance, and secondary alkyl radicals, which are stabilized through hyperconjugation. Among the seven resulting products, six exhibit optical activity, leading to the formation of a total of 13 possible isomers.
Approach Solution -2

The maximum number of possible isomers (including stereoisomers) formed on mono-bromination of 1-methylcyclohex-1-ene using Br2 and UV light is 13.
Here's the breakdown:
Number of Positions for Bromination: There are six carbons in the cyclohexene ring, so the bromine atom (Br) can be added to any of these six positions, resulting in six primary products.
Stereoisomers: The presence of a double bond and a methyl group introduces the possibility of stereoisomers. The double bond can be either cis or trans, and the methyl group can be on either side of the plane formed by the cyclohexene ring. However, not all positions will have both cis and trans isomers.
- Positions 2 and 5: These carbons are equivalent due to the symmetry of the molecule. Bromination at either position will result in the same product.
- Positions 1, 3, 4, and 6: These positions can lead to both cis and trans isomers with the methyl group, depending on the orientation of the double bond and the bromine atom.
Therefore, we can have:
- 2 positions with 1 isomer each (positions 2 and 5)
- 4 positions with 2 isomers each (positions 1, 3, 4, and 6)
Total isomers = 2 + (4 * 2) = 10
- Free Radical Mechanism: Bromination with Br2 and UV light involves a free radical mechanism. This mechanism can also lead to the formation of a racemic mixture for some of the isomers. A racemic mixture is a 1:1 mixture of enantiomers (mirror images that are not superimposable).
Therefore, for each of the 10 isomers with a chiral center (positions 1, 3, 4, and 6), we can have a racemic mixture, effectively doubling the number of possible stereoisomers.
Total isomers (including stereoisomers) = 10 + (4 * 2) = 18
- Ignoring Racemic Mixtures (Conservative Estimate): In some cases, the question might ask for the unique isomers, excluding racemic mixtures. Since we cannot definitively determine the presence or absence of racemic mixtures in this case, we can take a conservative approach and ignore them.
Therefore, the final answer is:
Maximum number of possible isomers (including stereoisomers): 13 (6 primary products + 7 stereoisomers)
Top Questions on Isomerism in Coordination Compounds
- Among V(CO)6, Cr(CO)5, Cu(CO)3, Mn(CO)5, Fe(CO)5, [Co(CO)3]3-, [Cr(CO)4]4-, and Ir(CO)3, the total number of species isoelectronic with Ni(CO)4 is _________.
[Given, atomic number: V = 23, Cr=24, Mn 25, Fe 26, Co 27, Ni 28, Cu = 29, Ir = 77]- JEE Advanced - 2024
- Chemistry
- Isomerism in Coordination Compounds
- The complex cation which has two isomers is :
- JEE Main - 2023
- Chemistry
- Isomerism in Coordination Compounds
- The $Cl - Co - Cl$ bond angle values in a fac- $\left[ Co \left( NH _3\right)_3 Cl _3\right]$ complex is/are:
- JEE Main - 2023
- Chemistry
- Isomerism in Coordination Compounds
- If Ni2– is replaced by Pt2– in the complex NiCl2Br2] 2– , which of the following properties are expected to get changed?
A. Geometry
B. Geometrical isomerism
C. Optical isomerism
D. Magnetic properties- JEE Main - 2023
- Chemistry
- Isomerism in Coordination Compounds
- L-isomer of a compound ‘A’ \((C_4H_8O_4)\) gives a positive test with \([Ag(NH_3)^2]^+\). Treatment of ‘A’ with acetic anhydride yields triacetate derivative. Compound ‘A’ produces an optically active compound (B) and an optically inactive compound (C) on treatment with bromine water and \(HNO_3\) respectively. Compound (A) is:
- JEE Main - 2022
- Chemistry
- Isomerism in Coordination Compounds
Questions Asked in JEE Advanced exam
- Let the function \(f:[1,\infin)→\R\) be defined by
\(f(t) = \begin{cases} (-1)^{n+1}2, & \text{if } t=2n-1,n\in\N, \\ \frac{(2n+1-t)}{2}f(2n-1)+\frac{(t-(2n-1))}{2}f(2n+1) & \text{if } 2n-1<t<2n+1,n\in\N. \end{cases}\)
Define \(g(x)=\int\limits_{1}^{x}f(t)dt,x\in(1,\infin).\) Let α denote the number of solutions of the equation g(x) = 0 in the interval (1, 8] and \(β=\lim\limits_{x→1+}\frac{g(x)}{x-1}\). Then the value of α + β is equal to _____.- JEE Advanced - 2024
- Integral Calculus
- A dimensionless quantity is constructed in terms of electronic charge \(e\), permittivity of free space \(\epsilon_0\) , Planck’s constant ℎ, and speed of light c. If the dimensionless quantity is written as \(e^\alpha\epsilon_0^\beta h^\gamma c^\delta\)and n is a non-zero integer, then\((\alpha, \beta,\gamma,\delta)\) is given by
- JEE Advanced - 2024
- Semiconductor electronics: materials, devices and simple circuits
- A block of mass \(5 kg\) moves along the \(x-\)direction subject to the force \(F = (−20x + 10) N,\) with the value of \(x \) in metre. At time \(t = 0 s,\) it is at rest at position \(x = 1 m\). The position and momentum of the block at \(t = (\pi/4)\) s are
- JEE Advanced - 2024
- Work-energy theorem
- A region in the form of an equilateral triangle (in x-y plane) of height L has a uniform magnetic field 𝐵⃗ pointing in the +z-direction. A conducting loop PQR, in the form of an equilateral triangle of the same height 𝐿, is placed in the x-y plane with its vertex P at x = 0 in the orientation shown in the figure. At 𝑡 = 0, the loop starts entering the region of the magnetic field with a uniform velocity 𝑣 along the +x-direction. The plane of the loop and its orientation remain unchanged throughout its motion.
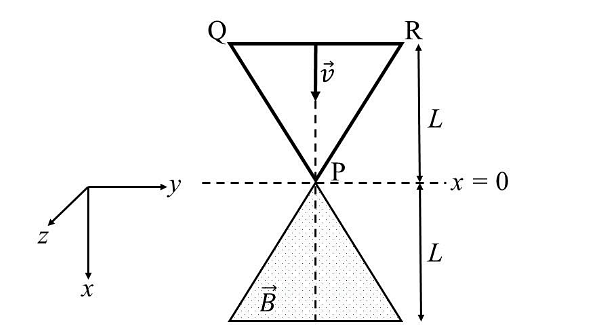
Which of the following graph best depicts the variation of the induced emf (E) in the loop as a function of the distance (𝑥) starting from 𝑥 = 0? - Two beads, each with charge q and mass m, are on a horizontal, frictionless, non-conducting, circular hoop of radius R. One of the beads is glued to the hoop at some point, while the other one performs small oscillations about its equilibrium position along the hoop. The square of the angular frequency of the small oscillations is given by [ \(\epsilon_0 \)is the permittivity of free space.]
- JEE Advanced - 2024
- Moving charges and magnetism
Concepts Used:
Isomerism
Isomerism
In simple words, multiple compounds having the same molecular formulae but different structural formulae are referred to as isomers and this entire phenomenon is described as ‘Isomerism’. ‘Isos’ and ‘meros’ are Greek words that translate to ‘equal parts’.
Types of Isomerism:
Isomerism can be differentiated by primary types and their subtypes.
- Structural Isomerism
Ring Chain Isomerism, Chain Isomerism, Tautomerism, Functional Isomerism, Metamerism, and Positional Isomerism are the subtypes of Structural Isomerism.
- Stereoisomerism
Optical Isomerism & Geometric Isomerism are the two subtypes of Stereoisomerism.
Read More: Isomerism in Coordination Compounds
Structural Isomerism
The phenomenon when the compounds start showing isomerism because of their different structure is termed Structural Isomerism. Example - Isobutane and n-butane
Structural Isomerism can further be classified into various types such as chain isomerism, positional isomerism, functional isomerism, metamerism, ring-chain isomerism, and tautomerism.
- Ring Chain isomerism
In the ring chain isomerism, there will be an open-chain structure in any one of the isomers and the remaining will have a closed chain or a ring structure. These kinds of compounds are known as ring chain isomers and this phenomenon is termed ring chain isomerism. The number of pi bonds in ring chain isomers will be different. Example - Cyclopropane and Propene. Their molecular formula is C3H6
- Chain isomerism
In chain isomerism, there will be different branches of carbon atoms in multiple isomers compounds but still, their molecular formula remains the same. Skeleton isomerism is the other term used to refer to chain isomerism. Example- 2 methyl butane, 2-2 dimethyl propane, and n-pentane. Their molecular formula is C5H12
- Tautomerism
In tautomerism, there will be a difference in isomer compounds only in the position of electrons, protons, and atoms. Tautomers take place in an equilibrium state as they can easily interchange. Example - Keto-enol tautomerism
- Functional Isomerism
In functional isomerism, the isomer compounds with different functional groups have the same molecular formula. Example - dimethyl ether and ethanol. They both have the same molecular formula C2H6 O. The functional group of Ethanol is ‘OH’ while the Functional group of dimethyl ether is ‘R-O-R’.
- Metamerism
In metamerism, each side of the functional group has the presence of different alkyl chains. Compared with other types of isomerism, metamerism is rarely found and is constrained to molecules with atoms. Example - Methoxy-propane (CH3OC3H7) & ethoxyethane (C2H5OC2H5)
- Positional Isomerism
In Positional isomerism, the functional groups and substituents of a structural isomer will have different positions. Example - 1-Chloropropane and 2-Chloropropane
Stereoisomerism
The phenomenon when compounds start showing different spatial arrangements of atoms even when their molecular formula is the same is termed Stereoisomerism. Example - 1,2 - dichloroethene (C2H2CI2)
- Optical isomerism
In optical isomerism, the isomer Compounds differ in terms of optical activity but are mirror images of each other. In this subtype of Stereoisomerism, carbon atoms are connected to four different groups and this is known as the chiral center. Enantiomers is the term mostly used to refer to optical isomers.
In Dextro optical isomers plane of polarized light is rotated to the right. In Laevo optical Isomers, the plane of polarized light is rotated to the left.
- Geometric isomerism
In geometric isomerism, the spatial positions of molecules are locked because of the presence of a double bond or ring structure. It is often referred to as Cis-Trans isomerism. Groups connected to ring structure carbon atom must be different in geometric isomerism.



