Match items of Column I with the items of Column II and assign the correct code:


- (a) $\to$ (ii) ,(b) $\to$ (iii) ,(c) $\to$ (i) ,(d) $\to$ (v)
- (a) $\to$ (i) ,(b) $\to$ (ii) ,(c) $\to$ (iii) ,(d) $\to$ (iv)
- (a) $\to$ (iii) ,(b) $\to$ (iv) ,(c) $\to$ (v) ,(d) $\to$ (i)
- (a) $\to$ (iv) ,(b) $\to$ (ii) ,(c) $\to$ (iii) ,(d) $\to$ (i)
The Correct Option is D
Approach Solution - 1
Extraction Processes
Cyanide Process:
The cyanide process is used to extract gold (Au) from its ores. The process involves the following reactions:
\(4Au + 8NaCN + O_2 + 2H_2O \to 4Na[Au(CN)_2] + 4NaOH\)
After this, the impure gold cyanide complex is treated with zinc to reduce the gold:
\(2Na[Au(CN)_2] \xrightarrow{[Zn]} \underset{\text{ppt}}{2 Au} + Na_2[Zn(CN)_4]\)
Froth Floatation Process:
The froth flotation process is used for concentrating sulphide ores. This method involves using air bubbles to separate the desired minerals from the rest of the ore by creating froth, where the mineral particles attach to the bubbles.
Electrolytic Reduction (Hall-Heroult Process):
The Hall-Heroult process is used to extract aluminium from its ore, bauxite. This process involves the electrolytic reduction of bauxite in the presence of cryolite:
Electrolytic reduction of bauxite is done by dissolving the ore in molten cryolite (Na₃AlF₆) and applying a direct current. This reduces aluminium oxide to aluminium metal at the cathode.
Zone Refining:
Zone refining is a method of purifying semiconductor materials like Germanium (Ge) and Silicon (Si). In this method, a molten zone is passed along the material, selectively dissolving impurities into the molten region, which is then moved to the next region for further purification.
Approach Solution -2
- Cyanide process: This process is used for the extraction of gold (Au), hence it is matched with (iv) Extraction of Au.
- Froth flotation process: This is used for concentrating sulphide ores like ZnS (zinc sulfide), so it is matched with (ii) Dressing of ZnS.
- Electrolytic reduction: This is the method used to extract aluminum (Al) from bauxite, so it is matched with (iii) Extraction of Al.
- Zone refining: This technique is used for purifying materials like germanium (Ge), so it is matched with (i) Ultrapure Ge.
Correct Code:
(a) → (iv), (b) → (ii), (c) → (iii), (d) → (i)
Top NEET Chemistry Questions
Which of the following statements are true?
A. Unlike Ga that has a very high melting point, Cs has a very low melting point.
B. On Pauling scale, the electronegativity values of N and C are not the same.
C. $Ar, K^{+}, Cl^{–}, Ca^{2+} and S^{2–}$ are all isoelectronic species.
D. The correct order of the first ionization enthalpies of Na, Mg, Al, and Si is Si $>$ Al $>$ Mg $>$ Na.
E. The atomic radius of Cs is greater than that of Li and Rb.
Choose the correct answer from the options given below:- NEET (UG) - 2025
- Chemistry
- Inorganic chemistry
- Given below are two statements:
Statement I: Like nitrogen that can form ammonia, arsenic can form arsine.
Statement II: Antimony cannot form antimony pentoxide.
In the light of the above statements, choose the most appropriate answer from the options given below:- NEET (UG) - 2025
- Chemistry
- Inorganic chemistry
For the reaction A(g) $\rightleftharpoons$ 2B(g), the backward reaction rate constant is higher than the forward reaction rate constant by a factor of 2500, at 1000 K.
[Given: R = 0.0831 atm $mol^{–1} K^{–1}$]
$K_p$ for the reaction at 1000 K is:- NEET (UG) - 2025
- Chemistry
- Law Of Chemical Equilibrium And Equilibrium Constant
Among the following, choose the ones with an equal number of atoms.
- 212 g of Na2CO3 (s) [molar mass = 106 g]
- 248 g of NaNO3 (s) [molar mass = 62 g]
- 240 g of NaOH (s) [molar mass = 40 g]
- 12 g of H2 (g) [molar mass = 2 g]
- 220 g of CO2 (g) [molar mass = 44 g]
Choose the correct answer from the options given below:
- NEET (UG) - 2025
- Chemistry
- Stoichiometry and Stoichiometric Calculations
Identify the suitable reagent for the following conversion: $Ph-C(=O)-OCH_3$ $\longrightarrow$ $Ph-CHO$
- NEET (UG) - 2025
- Chemistry
- Organic Chemistry
Top NEET General Principles and Processes of Isolation of Elements Questions
The reaction that does NOT take place in a blast furnace between 900 K to 1500 K temperature range during extraction of iron is?
- NEET (UG) - 2023
- Chemistry
- General Principles and Processes of Isolation of Elements
- Which one of the following methods can be used to obtain highly pure metal which is liquid at room temperature?
- NEET (UG) - 2021
- Chemistry
- General Principles and Processes of Isolation of Elements
- Which one is malachite from the following ?
- NEET (UG) - 2019
- Chemistry
- General Principles and Processes of Isolation of Elements
- Considering Ellingham diagram, which of the following metals can be used to reduce alumina ?
- NEET (UG) - 2018
- Chemistry
- General Principles and Processes of Isolation of Elements
- Extraction of gold and silver involes leaching with $CN^-$ ion. Silver is later recovered by :-
- NEET (UG) - 2017
- Chemistry
- General Principles and Processes of Isolation of Elements
Top NEET Questions
- Which of the following enzyme(s) are NOT essential for gene cloning?
A. Restriction enzymes
B. DNA ligase
C. DNA mutase
D. DNA recombinase
E. DNA polymerase- NEET (UG) - 2025
- Molecular Biology
A constant voltage of 50 V is maintained between the points A and B of the circuit shown in the figure. The current through the branch CD of the circuit is :

- NEET (UG) - 2025
- Current electricity
- Frogs respire in water by skin and buccal cavity and on land by skin, buccal cavity and lungs.
- NEET (UG) - 2025
- Animal Physiology
Which of the following statements are true?
A. Unlike Ga that has a very high melting point, Cs has a very low melting point.
B. On Pauling scale, the electronegativity values of N and C are not the same.
C. $Ar, K^{+}, Cl^{–}, Ca^{2+} and S^{2–}$ are all isoelectronic species.
D. The correct order of the first ionization enthalpies of Na, Mg, Al, and Si is Si $>$ Al $>$ Mg $>$ Na.
E. The atomic radius of Cs is greater than that of Li and Rb.
Choose the correct answer from the options given below:- NEET (UG) - 2025
- Inorganic chemistry
AB is a part of an electrical circuit (see figure). The potential difference \(V_A - V_B\), at the instant when current \(i = 2\) A and is increasing at a rate of 1 amp/second is:

- NEET (UG) - 2025
- Electromagnetic Induction and Inductance
Concepts Used:
General Principles and Processes of Isolation of Elements
What are Ores and Minerals?
Minerals are the naturally occurring, homogeneous inorganic solid substances. They are having a definite chemical composition and crystalline structure, hardness and color. For example, copper pyrite, calamine, etc.
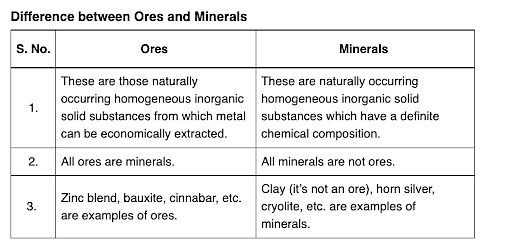
Impurities in an ore are called gauge. The removal of a gauge from the ore is called concentration ore.
Several steps are involved in the extraction of pure metal from ores. Major steps are as follows –
- Concentration of the ore
- Isolation of the metal from its concentrated ore
- Purification of the metal