Consider the following statements about the oxides of halogens
A. At room temperature, OF2; is thermally stable
B. Order of stability of oxides of halogens is I > Br > Cl
C. I2O5 is used in the estimation of CO
D. ClO2; is used as a bleaching agent
The correct statements are
Consider the following statements about the oxides of halogens
A. At room temperature, OF2; is thermally stable
B. Order of stability of oxides of halogens is I > Br > Cl
C. I2O5 is used in the estimation of CO
D. ClO2; is used as a bleaching agent
The correct statements are
A & B only
A,B & C only
B & C only
A,C & D only
The Correct Option is D
Solution and Explanation
The correct option is: (D): A, C & D only
Top Questions on General Principles and Processes of Isolation of Elements
The reaction that does NOT take place in a blast furnace between 900 K to 1500 K temperature range during extraction of iron is?
- NEET (UG) - 2023
- Chemistry
- General Principles and Processes of Isolation of Elements
- Which of the following ore is not in oxide form?
- GUJCET - 2023
- Chemistry
- General Principles and Processes of Isolation of Elements
- Which of the following shows an incorrect method of refining?
- JEE Main - 2023
- Chemistry
- General Principles and Processes of Isolation of Elements
- The metal which is extracted by oxidation and subsequent reduction from its ore is:
- JEE Main - 2023
- Chemistry
- General Principles and Processes of Isolation of Elements
- Given below are two statements:
Statement I : During Electrolytic refining, the pure metal is made to act as anode and its impure metallic form is used as cathode
Statement II : During the Hall-Heroult electrolysis process, purified $Al _2 O _3$ is mixed with $Na _3 AlF _6$ to lower the melting point of the mixture
In the light of the above statements, choose the most appropriate answer from the options given below:- JEE Main - 2023
- Chemistry
- General Principles and Processes of Isolation of Elements
Questions Asked in TS EAMCET exam
The number of significant figures in the measurement of a length 0.079000 m is:
- TS EAMCET - 2023
- Units and measurement
The roots of the equation x4 + x3 - 4x2 + x + 1 = 0 are diminished by h so that the transformed equation does not contain x2 term. If the values of such h are α and β, then 12(α - β)2 =
- TS EAMCET - 2023
- Quadratic Equations
The number of electrons with (n+1) values equal to 3,4 and 5 in an element with atomic number (z) 24 are respectively (n = principal quantum number and l = azimuthal quantum number)
- TS EAMCET - 2023
- atom structure models
Two convex lenses of focal lengths 20 cm and 30 cm are placed in contact with each other co-axially. The focal length of the combination is:
- TS EAMCET - 2023
- Ray optics and optical instruments
- If the ratio of densities of two substances is 5:6 and the ratio of their specific heat capacities is 3:5, then the ratio of heat energies required per unit volume so that the two substances can have same temperature rise is:
- TS EAMCET - 2023
- Thermodynamics
Concepts Used:
General Principles and Processes of Isolation of Elements
What are Ores and Minerals?
Minerals are the naturally occurring, homogeneous inorganic solid substances. They are having a definite chemical composition and crystalline structure, hardness and color. For example, copper pyrite, calamine, etc.
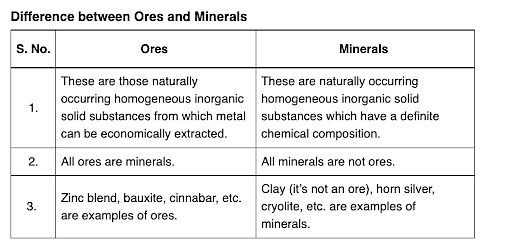
Impurities in an ore are called gauge. The removal of a gauge from the ore is called concentration ore.
Several steps are involved in the extraction of pure metal from ores. Major steps are as follows –
- Concentration of the ore
- Isolation of the metal from its concentrated ore
- Purification of the metal



