Question:
The stretching frequency of CO in H3B.CO is
The stretching frequency of CO in H3B.CO is
Updated On: Aug 13, 2024
- greater than the stretching frequency in free CO.
- lesser than the stretching frequency in free CO.
- lesser than the stretching frequency of CO in Fe(CO)5.
- greater than the stretching frequency of CO in Fe(CO)5.
Hide Solution
Verified By Collegedunia
The Correct Option is A, D
Solution and Explanation
The correct options are: A and D
Was this answer helpful?
0
0
Top Questions on Spectroscopy
- The wavelength of the γ-ray emitted in
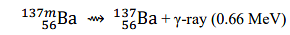
is _______ Å. (rounded off to three decimal places)
[Given: h=6.626×10 −34 J s; c=2.998×108 m s−1 ; 1 MeV=1.602×10−13 J]- IIT JAM CY - 2024
- Physical Chemistry
- Spectroscopy
- If 1 M of a dye in water transmits 50% of incident light at 400 nm, then 2 M of the dye in water transmits _______ % of the incident light at 400 nm. (rounded off to the nearest integer)
[Given: Both experiments are performed in the same spectrophotometric cell.]- IIT JAM CY - 2024
- Physical Chemistry
- Spectroscopy
- For the following compound
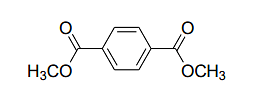
the number of signals expected in the 1H NMR spectrum is _________.- IIT JAM CY - 2024
- Physical Chemistry
- Spectroscopy
- The harmonic vibrational frequency of a diatomic molecule is 2000 cm−1 . Its zero point energy is eV.
[Given: Planck’s constant = 6.62x10−34 J s; 1 eV = 1.6x10−19 J]
(round off to two decimal places)- IIT JAM CY - 2023
- Physical Chemistry
- Spectroscopy
- The highest possible energy of a photon in the emission spectrum of hydrogen atom is ______ eV.
[Given: Rydberg constant = 13.61 eV]
(round off to two decimal places)- IIT JAM CY - 2023
- Physical Chemistry
- Spectroscopy
View More Questions
Questions Asked in IIT JAM CY exam
- The number and nature of d−d transition(s) in the case of Sc2+ in an octahedral crystal field, respectively, are
[Ignore spin-orbit coupling and Jahn-Teller distortion.]- IIT JAM CY - 2024
- d block elements
- A system undergoes one clockwise cycle from point X back to point X as shown in the figure below:
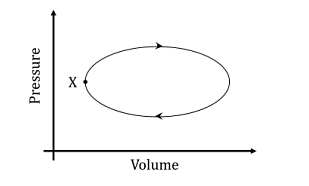
The correct statement about this process is- IIT JAM CY - 2024
- Thermodynamics
- A salt QCl of a certain metal Q is electrolyzed to its elements. 40 g of metal Q is formed at an electrode. The volume of Cl2 formed at the other electrode at 1 atm pressure and 298 K is _______ litres. (rounded off to one decimal place)
[Given: The gas constant 𝑅 = 0.082 L atm mol−1 K−1 , the molar mass of Q is 40 g mol−1 and Cl2 is assumed to be an ideal gas]- IIT JAM CY - 2024
- Electrochemistry
- Consider photoelectric effect. The number of incident photons is the same for all frequencies. The plot that best describes the dependence of the number of photoelectrons (n) emitted as a function of the incident light frequency (ν) is
- IIT JAM CY - 2024
- Atomic Structure
- The major product formed in the following reaction
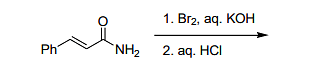
is- IIT JAM CY - 2024
- Reaction Mechanisms & Synthesis
View More Questions



