A 12.5 eV electron beam is used to bombard gaseous hydrogen at room temperature. The number of spectral lines emitted will be
- 1
- 2
- 3
- 4
The Correct Option is C
Solution and Explanation
According to Bohr's postulates, an electron makes a jump to higher energy orbital if it absorbs a photon of energy equal to the difference between the energies of an excited state and the ground state. Assuming that the collided electron takes energy equal to 10.2 eV or 12.09 eV from the incoming electron beam (some part lost due to collision), the maximum excited state is \( n = 3 \). The number of spectral lines is given by: \[ \frac{3(3 - 1)}{2} = 3. \] Thus, the number of spectral lines emitted is 3.
Top JEE Main Physics Questions
- Water drops fall from a tap on the floor, \(5\) m below, at regular intervals of time. The first drop strikes the floor when the sixth drop begins to fall. The height at which the fourth drop will be from the ground, at the instant when the first drop strikes the ground, is ________ m. (\( g = 10 \, \text{m s}^{-2} \))
- JEE Main - 2026
- Physics
- Kinematics
- A block of mass \(5\) kg is moving on an inclined plane which makes an angle of \(30^\circ\) with the horizontal. The coefficient of friction between the block and the inclined plane surface is \(\dfrac{\sqrt{3}}{2}\). The force to be applied on the block so that the block moves {down the plane without acceleration is ________ N. (\( g = 10 \, \text{m s}^{-2} \))}
- JEE Main - 2026
- Physics
- Newtons Laws of Motion
Identify the correct truth table of the given logic circuit.

- JEE Main - 2026
- Physics
- communication systems
- The escape velocity from a spherical planet \(A\) is \(10\ \text{km/s}\). The escape velocity from another planet \(B\), whose density and radius are \(10%\) of those of planet \(A\), is _______ m/s.
- JEE Main - 2026
- Physics
- Gravitation
- A current carrying solenoid is placed vertically and a particle of mass m with charge Q is released from rest. The particle moves along the axis of solenoid. If g is acceleration due to gravity then the acceleration (a) of the charged particle will satisfy:
- JEE Main - 2026
- Physics
- Moving charges and magnetism
Top JEE Main specific heat capacity Questions
Match List-I with List-II.
List-I List-II (A) Heat capacity of body (I) \( J\,kg^{-1} \) (B) Specific heat capacity of body (II) \( J\,K^{-1} \) (C) Latent heat (III) \( J\,kg^{-1}K^{-1} \) (D) Thermal conductivity (IV) \( J\,m^{-1}K^{-1}s^{-1} \) - JEE Main - 2025
- Physics
- specific heat capacity
- Two moles a monoatomic gas is mixed with six moles of a diatomic gas. The molar specific heat of the mixture at constant volume is :
- JEE Main - 2024
- Physics
- specific heat capacity
The pressure of a gas changes linearly with volume from $A$ to $B$ as shown in figure If no heat is supplied to or extracted from the gas then change in the internal energy of the gas will be Is
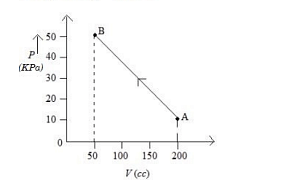
- JEE Main - 2023
- Physics
- specific heat capacity
Let \(\gamma_1\)be the ratio of molar specific heat at constant pressure and molar specific heat at constant volume of a monoatomic gas and \(\gamma_2\) be the similar ratio of diatomic gas. Considering the diatomic gas molecule as a rigid rotator, the ratio, \(\frac{\gamma_1}{\gamma_2}\) is :
- JEE Main - 2023
- Physics
- specific heat capacity
A water heater of power $2000 W$ is used to heat water. The specific heat capacity of water is $4200 J$ $kg ^{-1} K ^{-1}$ .The efficiency of heater is $70 \%$ .Time required to heat $2 kg$ of water from $10^{\circ} C$ to $60^{\circ} C$ is___$ s$
(Assume that the specific heat capacity of water remains constant over the temperature range of the water)- JEE Main - 2023
- Physics
- specific heat capacity
Top JEE Main Questions
- Let $a_1,a_2,a_3,a_4$ be an A.P. of four terms such that each term of the A.P. and its common difference are integers. If $a_1+a_2+a_3+a_4=48$ and $a_1^2a_2a_3a_4+1^4=361$, then the largest term of the A.P. is equal to
- JEE Main - 2026
- Arithmetic Progression
- If the coefficient of \( x \) in the expansion of \[ (ax^2 + bx + c)(1 - 2x)^{26} \] is \(-56\) and the coefficients of \( x^2 \) and \( x^3 \) are both zero, then \( a + b + c \) is equal to
- JEE Main - 2026
- Binomial theorem
- Water drops fall from a tap on the floor, \(5\) m below, at regular intervals of time. The first drop strikes the floor when the sixth drop begins to fall. The height at which the fourth drop will be from the ground, at the instant when the first drop strikes the ground, is ________ m. (\( g = 10 \, \text{m s}^{-2} \))
- JEE Main - 2026
- Kinematics
- Let \( y = x \) be the equation of a chord of the circle \( C_1 \) (in the closed half-plane \( x \ge 0 \)) of diameter 10 passing through the origin. Let \( C_2 \) be another circle described on the given chord as diameter. If the equation of the chord of the circle \( C_2 \), which passes through the point \( (2, 3) \) and is farthest from the center of \( C_2 \), is \( x + ay + b = 0 \), then \( b \) is equal to:
- JEE Main - 2026
- Coordinate Geometry
- If \[ \frac{\tan(A-B)}{\tan A}+\frac{\sin^2 C}{\sin^2 A}=1, \quad A,B,C\in\left(0,\frac{\pi}{2}\right), \] then:
- JEE Main - 2026
- Trigonometry