Question:
Order of magnitude of density of uranium nucleus is
$ (m_p = 1.67 \times 10^{-27} Kg ) $
Order of magnitude of density of uranium nucleus is
$ (m_p = 1.67 \times 10^{-27} Kg ) $
Updated On: Aug 15, 2022
- $ 10^{20} Kg/m^3 $
- $ 10^{17} Kg/m^3 $
- $ 10^{14} Kg/m^3 $
- $ 10^{11} Kg/m^3 $
Hide Solution
Verified By Collegedunia
The Correct Option is B
Solution and Explanation
Radius of a nucleus is given by
$ R = R_0 A^{1/3} (Where R_0 = 1.25 \times 10^{-15} m ) $
$ = 1.25 A^{1/3} \times 10^{-15} m $
Here A is the mass number and mass of the uranium nucleus
will be
$ m = Am_p $ where $ m_p = mass \, of \, proton $
$ = A (1.67 \times 10^{-27} Kg ) $
$ \therefore $ Density $ p = \frac{mass}{volume} = \frac{m}{\frac{4}{3} \pi R^3} $
$ = \frac{A(1.67 \times 10^{-27} Kg)}{A(1.25 \times 10^{-15}m)^3} \, \, or \, \, p \approx 2.0 \times 10^{17} Kg/m^3 $
Was this answer helpful?
0
0
Top Questions on Nuclei
- The disintegration energy \( Q \) for the nuclear fission of \( ^{235}\text{U} \to ^{140}\text{Ce} + ^{94}\text{Zr} + n \) is _____ MeV.
Given atomic masses:
\[^{235}\text{U} = 235.0439 \, u, \quad ^{140}\text{Ce} = 139.9054 \, u, \quad ^{94}\text{Zr} = 93.9063 \, u, \quad n = 1.0086 \, u\]Value of \( c^2 = 931 \, \text{MeV/u} \). - The mass defect in a particular reaction is \( 0.4 \, \text{g} \). The amount of energy liberated is \( n \times 10^7 \, \text{kWh} \), where \( n = \) _____. (speed of light \( = 3 \times 10^8 \, \text{m/s} \))
- A nucleus has mass number \(A_1\) and volume \(V_1\). Another nucleus has mass number \(A_2\) and volume \(V_2\). If the relation between mass numbers is \(A_2 = 4A_1\), then \(\frac{V_2}{V_1} =\) _______.
- The mass number of nucleus having radius equal to half of the radius of nucleus with mass number 192 is:
- A nucleus at rest disintegrates into two smaller nuclei with their masses in the ratio of 2:1. After disintegration they will move
View More Questions
Questions Asked in JEE Advanced exam
- Let the function \(f:[1,\infin)→\R\) be defined by
\(f(t) = \begin{cases} (-1)^{n+1}2, & \text{if } t=2n-1,n\in\N, \\ \frac{(2n+1-t)}{2}f(2n-1)+\frac{(t-(2n-1))}{2}f(2n+1) & \text{if } 2n-1<t<2n+1,n\in\N. \end{cases}\)
Define \(g(x)=\int\limits_{1}^{x}f(t)dt,x\in(1,\infin).\) Let α denote the number of solutions of the equation g(x) = 0 in the interval (1, 8] and \(β=\lim\limits_{x→1+}\frac{g(x)}{x-1}\). Then the value of α + β is equal to _____.- JEE Advanced - 2024
- Integral Calculus
- A dimensionless quantity is constructed in terms of electronic charge \(e\), permittivity of free space \(\epsilon_0\) , Planck’s constant ℎ, and speed of light c. If the dimensionless quantity is written as \(e^\alpha\epsilon_0^\beta h^\gamma c^\delta\)and n is a non-zero integer, then\((\alpha, \beta,\gamma,\delta)\) is given by
- JEE Advanced - 2024
- Semiconductor electronics: materials, devices and simple circuits
- A block of mass \(5 kg\) moves along the \(x-\)direction subject to the force \(F = (−20x + 10) N,\) with the value of \(x \) in metre. At time \(t = 0 s,\) it is at rest at position \(x = 1 m\). The position and momentum of the block at \(t = (\pi/4)\) s are
- JEE Advanced - 2024
- Work-energy theorem
- A region in the form of an equilateral triangle (in x-y plane) of height L has a uniform magnetic field 𝐵⃗ pointing in the +z-direction. A conducting loop PQR, in the form of an equilateral triangle of the same height 𝐿, is placed in the x-y plane with its vertex P at x = 0 in the orientation shown in the figure. At 𝑡 = 0, the loop starts entering the region of the magnetic field with a uniform velocity 𝑣 along the +x-direction. The plane of the loop and its orientation remain unchanged throughout its motion.
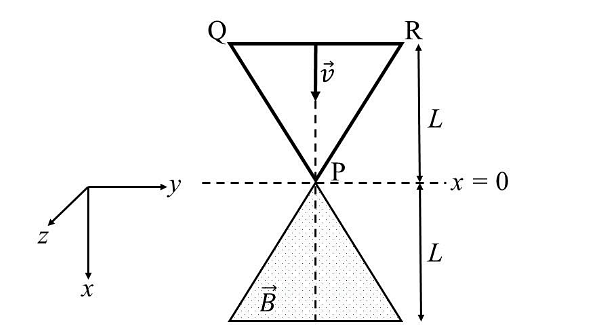
Which of the following graph best depicts the variation of the induced emf (E) in the loop as a function of the distance (𝑥) starting from 𝑥 = 0? - Two beads, each with charge q and mass m, are on a horizontal, frictionless, non-conducting, circular hoop of radius R. One of the beads is glued to the hoop at some point, while the other one performs small oscillations about its equilibrium position along the hoop. The square of the angular frequency of the small oscillations is given by [ \(\epsilon_0 \)is the permittivity of free space.]
- JEE Advanced - 2024
- Moving charges and magnetism
View More Questions
Concepts Used:
Nuclei
In the year 1911, Rutherford discovered the atomic nucleus along with his associates. It is already known that every atom is manufactured of positive charge and mass in the form of a nucleus that is concentrated at the center of the atom. More than 99.9% of the mass of an atom is located in the nucleus. Additionally, the size of the atom is of the order of 10-10 m and that of the nucleus is of the order of 10-15 m.
Read More: Nuclei
Following are the terms related to nucleus:
- Atomic Number
- Mass Number
- Nuclear Size
- Nuclear Density
- Atomic Mass Unit



